Video
I came across the original video of the seagull meme!
What a glorious day! I never realized it was a video, let alone a full throat and hearty evil villain laugh!
137K notes
·
View notes
Text








—Talk Ville Podcast + Smallville, “X-Ray”
176 notes
·
View notes
Text


My clex doodles
47 notes
·
View notes
Text

“I think I understand a whole lot better than you do”
77K notes
·
View notes
Text
Please check out my Good Omens art!!
0 notes
Link
Chapters: 1/1
Fandom: Good Omens (TV), Good Omens - Neil Gaiman & Terry Pratchett
Rating: Teen And Up Audiences
Warnings: No Archive Warnings Apply
Relationships: Aziraphale/Crowley (Good Omens)
Characters: Aziraphale (Good Omens), Crowley (Good Omens)
Additional Tags: Panic Attacks, Confessions
Summary:
Aziraphale is panicked by something rather serious and Crowley attempts to calm his friend but gets more than he'd bargained for.
0 notes
Text
When sodium hypochlorite (bleach) solution is added to luminol, a chemical reaction occurs that releases energy in the form of light. This is called chemiluminescence. The bleach solution acts as an oxidizing agent, which means it takes electrons away from the luminol molecule. This causes the luminol molecule to become excited, and it releases the energy as light.
🎥 Courtesy: Kendra Frederick
The luminol molecule is made up of two amino groups, a carbonyl group, and an azo group. The amino groups are electron-rich, while the carbonyl group is electron-poor. The azo group is a conjugated system, which means that the electrons in the double bonds can move freely from one atom to another.
When sodium hypochlorite (bleach) solution is added to luminol, the bleach molecules react with the amino groups of the luminol molecule. This reaction takes electrons away from the luminol molecule, which causes the luminol molecule to become oxidized. The oxidized luminol molecule is in an excited state, which means that it has more energy than it normally does.
The excited luminol molecule then releases the extra energy as light. This light is called chemiluminescence. The light emitted by the chemiluminescence reaction is blue because the luminol molecule has a blue fluorescence.
The chemiluminescence reaction between luminol and sodium hypochlorite is catalyzed by the presence of a metal ion, such as iron or copper. The metal ion helps to stabilize the excited state of the luminol molecule, which makes it more likely to release the extra energy as light.
The chemiluminescence reaction is very sensitive to impurities, so it is important to use pure chemicals. The reaction can also be affected by the pH of the solution. The optimal pH for the reaction is around 9.
The chemiluminescence reaction between luminol and sodium hypochlorite can be used to detect blood, as the iron in hemoglobin can catalyze the reaction. The reaction is also used in some commercial products, such as glow sticks and emergency lights.
I hope you enjoyed learning about this. ❤️🙏
5K notes
·
View notes
Text
Sneaky mom, she just got herself a infinite supply of serotonin
107 notes
·
View notes
Text

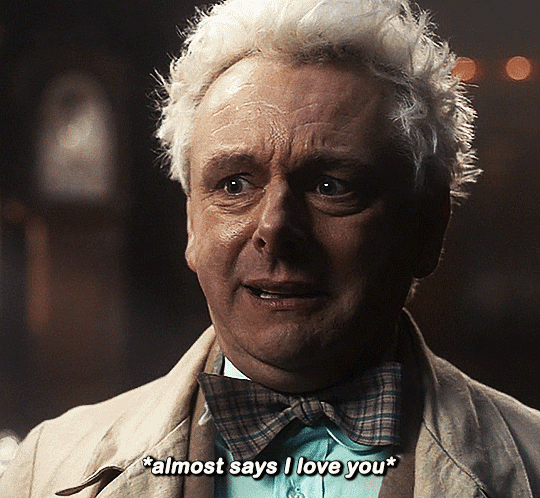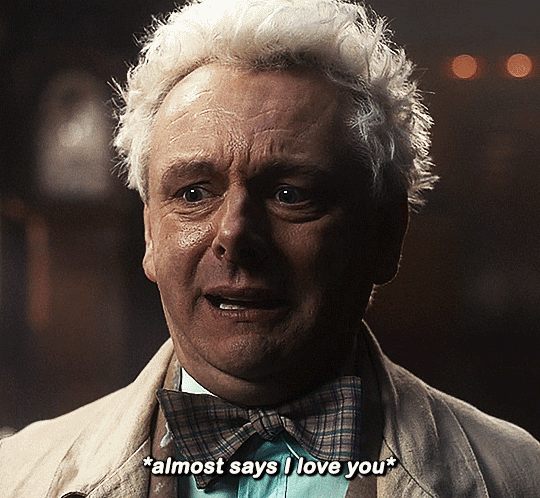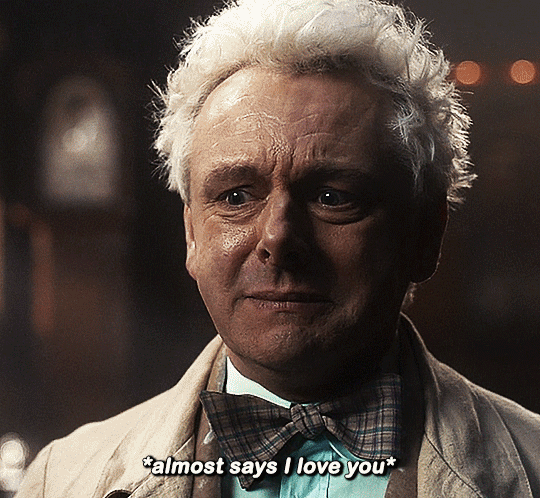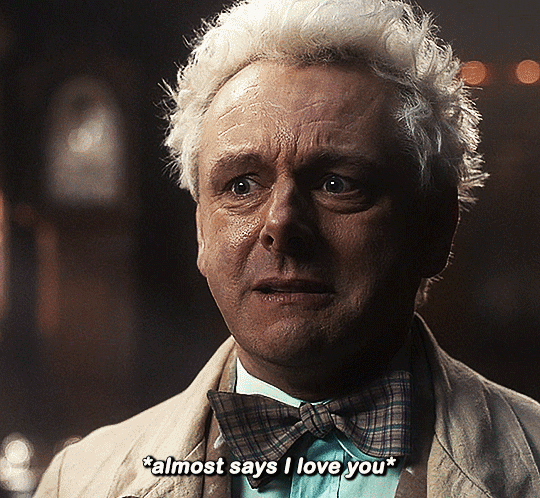
#Aziraphale didn’t know he was starving for Crowley’s love until he tasted him.

37K notes
·
View notes
Text






Good Omens | 2.06 -"Every Day" | Crowley/Aziraphale Kiss
You idiot. We could have been...us.
I...I forgive you.
6K notes
·
View notes
Text
Action Comics #1050
a.k.a. finally, some good fucking Superman and Lex content. This is the first issue in a long time where Lex has been allowed to be messy and human, AND have some kind of emotional continuity. Naturally, it’s because of Clark. Smallville fans, rejoice!
So ya boi has been doing some pondering after almost destroying the universe:

If you recall, this was the same pitch Lex used when he was trying to be Bruce Wayne’s Bat-themed sugar daddy. Apparently, the constant near-destruction of the multiverse really has warmed Lex up to the idea of superheroes.

Lex expressed some very similar feelings back when he took up the mantle of Superman. (If you’re wondering why Lex stopped trying to be a hero in the first place, the answer is very stupid.) But hey, this seems positive?

…oh. Peep the Kryptonite green shade of ‘my Superman.’

Some background: Up until this issue, the entire world knew that Clark (and also Jon) Kent was Superman. They were officially out of the super-closet. This issue begins with Perry White coming to dinner at the Kent Farm, saying that he has no idea who Superman’s secret identity is, and literally having a stroke at the revelation. Lex reveals that the mass memory wipe is his doing and is permanent, and takes Clark’s objections very calml–just kidding, he immediately gets in his Warsuit and starts punching.

I’ve read fanfic where Lex was keeping Clark in a cage that was still less possessive than this panel. Anyway, Clark punches Lex all the way onto the moon, and we get into the actual reason why Lex is doing all of this:



*screams in Smallville*
256 notes
·
View notes
Photo

Shout out to artist David Nakayama for capturing the true essence of Lex’s mood whenever Superman comes to visit/yell at him in jail: Smug Knife Cat

319 notes
·
View notes
Photo
Lovers


Clark Kent & Lex Luthor in “Drone”
742 notes
·
View notes