#tetrahedron
Text
SPAWN OF AZATHOTH
by Jakub Bazyluk

#tentacles#fhtagn#jakub bazyluk#machina arcana#lovecraft#cthulhu#azathoth#spawn#tetrahedron#concept art#floating#creature#monster#horror#fhtagnnn
382 notes
·
View notes
Text



tetrahedron gears fidget toy | source
#minecraft vibes. a little bit#talos gifs#stim gifs#stim#stim toys#fidget toys#body stim#irl hands#geometric#shapes#triangles#prism#tetrahedron#math#does this count. sure#mechanical#gears#plastic#blue#green#black#purple#dark green#teal#gif ids#id in alt
69 notes
·
View notes
Text

Quick friend-shapes needed to be made.
(meaning: I was bored and needed to do something with my hands and brain.)
82 notes
·
View notes
Text
Regular-ish Convex Polyhedra Bracket — Round 3
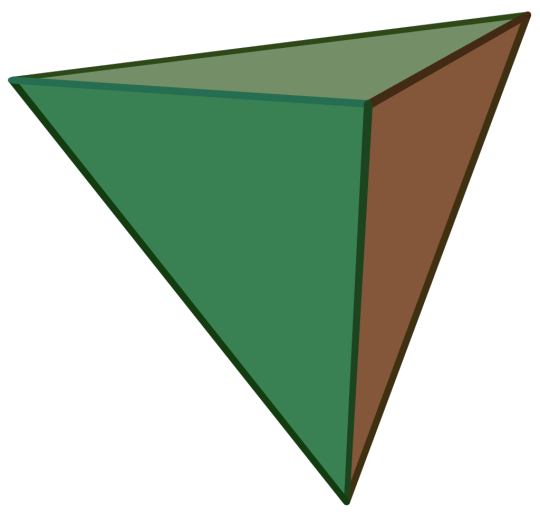
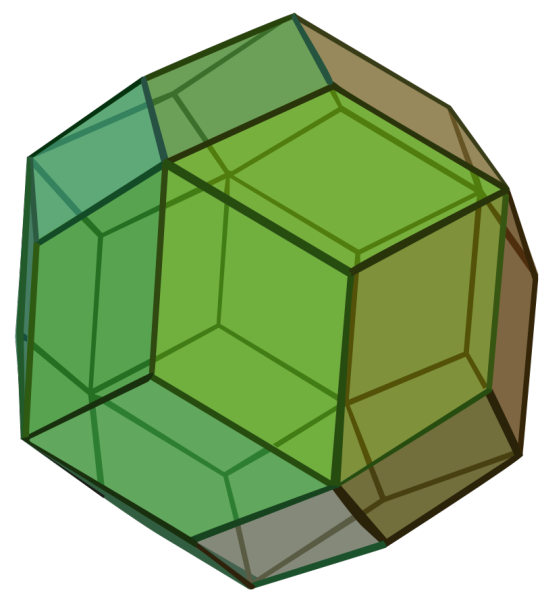
Propaganda
Regular Tetrahedron:
Also called the Triangular Pyramid
Platonic Solid
Regular
Dual of the Regular Tetrahedron
It has 4 regular triangular faces, 6 edges, and 4 vertices.
Self-Dual
Image Credit: Cyp
Rhombic Triacontahedron:
Also called the Triacontahedron
Catalan Solid
Dual of a quasiregular polyhedron
Dual of the Icosidodecahedron
It has 30 rhombic faces, 62 edges, and 32 vertices of two types.
One of the 9 edge-transitive convex polyhedra along with the 5 Platonic Solids, the 2 Quasiregular Convex Polyhedra, and the Rhombic Dodecahedron.
Image Credit: Maxim Razin based on Cyp
#Round 3#Regular Tetrahedron#Tetrahedron#Rhombic Triacontahedron#Platonic Solids#Catalan Solids#Polyhedra
60 notes
·
View notes
Text
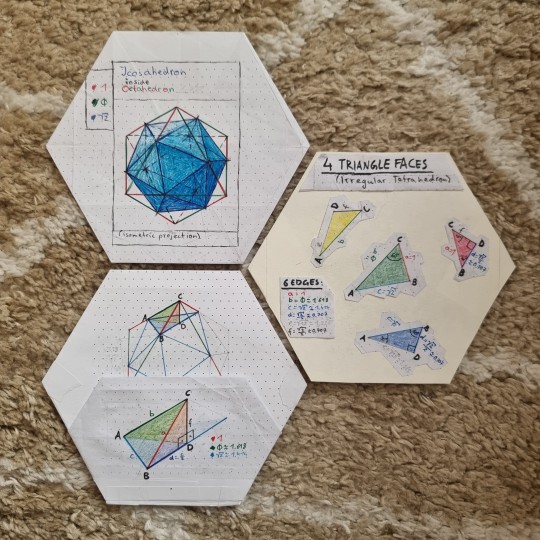
A visual study of the Icosahedron inside an Octahedron - and the remaining tetrahedral modules
The tetrahedral modules:
Buckminster Fuller came up with these modules and called them "S modules" or "S quanta modules".
Here is a helpful summary:

[Source (PDF) | A Fuller Explanation: The Synergetic Geometry of R. Buckminster Fuller, 1987]
#polyhedra#polyhedron#buckminster fuller#s-modules#modules#geometry#icosahedron#octahedron#platonic solids#tetrahedron#irregular tetrahedron#math art#hexagon#hexagrid#hexagon grid#mathy art#isometric#isometric projection#isometry
43 notes
·
View notes
Text
Since people are enjoying my slab bowl video so much, here's a video of me making The Shape:
Don't know what exactly to call it, but it brings me joy

106 notes
·
View notes
Text
#platonic solids#geometry#math#maths#science#shapes#poll#tetrahedron#cube#octahedron#dodecahedron#icosahedron
31 notes
·
View notes
Text









Trans tetrahedron.
Proof of concept.
Would like to make one out of wood and make a paint dip jig to get the vertex coloring level.
Think I need to do some math first and color blue and pink based on equal area, instead of simply starting equidistant along the edges.
2 notes
·
View notes
Text

I can feel the steel strangling the earth
I send silver
3 notes
·
View notes
Text
This permits every water molecule to be involved in four hydrogen bonds to four other H2O partners, as is shown in figure 6.41a. Hydrogen bonding in ice creates a three-dimensional network (figure 6.41b) that puts each oxygen atom at the centre of a distorted tetrahedron.
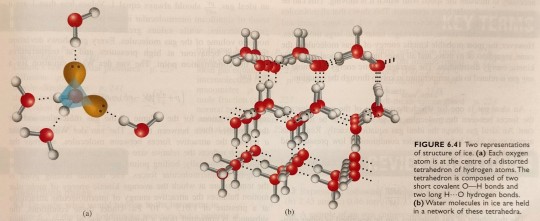
"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
4 notes
·
View notes
Text

A Chart of my Polyhedra JSAB OC's
#jsab#just shapes and beats#jsab oc#Pyrope#Tetrahedron#Octahedron#Dodecahedron#Icosahedron#Cuboctahedron#Rhombicuboctahedron#Rhombic Dodecahedron#Deltoidal Icositetrahedron#Snub Disphenoid#Triaugmented Triangular Prism#Gyroelongated Square Bipyramid#Gyrobifastigium
4 notes
·
View notes
Text



Today's Tiny Tetrahedron
(artist of the colorful artwork in the background of the first photo is unknown)
#tetrahedron#tetrahedra#platonic solid#platonic solids#polyhedra#polyhedron#geometry#mathy stuffy#art#math art#mathy art#crafts#my crafts#crafty#living in cubes....#no wait...#living in platonic solids#tetrahome#tetrahedra-octahedra honeycomb#space filling
100 notes
·
View notes
Text
Regular-ish Convex Polyhedra Bracket — Round 2
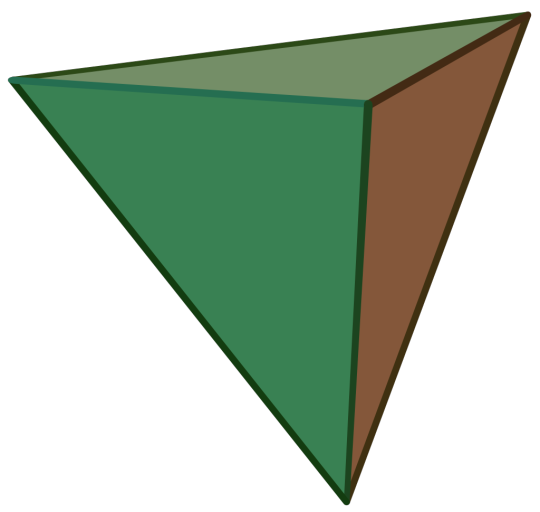

Propaganda
Regular Tetrahedron:
Also called the Triangular Pyramid
Platonic Solid
Regular
Dual of the Regular Tetrahedron
It has 4 regular triangular faces, 6 edges, and 4 vertices.
Self-Dual
Image Credit: Cyp
Pentagonal Icositetrahedron:
Also called the Pentagonal Icosikaitetrahedron
Catalan Solid
Dual of the Snub Cube
It has 24 irregular pentagonal faces, 60 edges, and 38 vertices of three types.
Chiral so it has two forms that are mirror images of each other.
Image Credit: Cyp
#Round 2#Regular Tetrahedron#Tetrahedron#Pentagonal Icositetrahedron#Platonic Solids#Catalan Solids#Polyhedra
13 notes
·
View notes
Text


1st iteration of the Sierpinski Tetrahedron - with an octahedron at the center.
The octahedron serves as "core".
Attached to that octahedron core are 4 tetrahedra.
Modules used: 24
[ 12 for the octahedron, 4x3 for the 4 tetrahedral tips ]
If I attach 4 more tetrahedral tips - so that all faces of the octahedron are stellated - it would turn into a stellated octahedron/stella octangula.
#origami#papee folding#paper crafts#paper folding#tetrahedron#tetrahedra#octahedron#polyhedra#polyhedron#geometry#math#mathy#mathy crafts#shapes#fractal#sierpinski tetrahedron#sierpinski#selfsimilarity
59 notes
·
View notes
Link
“An international collaboration of scientists has created and observed an entirely new class of vortices—the whirling masses of fluid or air.
Led by researchers from Amherst College in the U.S. and the University of East Anglia and Lancaster University in the U.K., their new paper details the first laboratory studies of these "exotic" whirlpools in an ultracold gas of atoms at temperatures as low as tens of billionths of a degree above absolute zero.”
“The unusual nature of the observed whirlpools here, however, is due to symmetries in the quantum gas. One especially fascinating property of physical theories, from cosmology to elementary particles, is the appearance of asymmetric worlds despite perfect underlying symmetries. For example, when water freezes to ice, disordered molecules in a liquid arrange themselves into a periodic array.
The spatial symmetry of a system is often readily identified—for example, a honeycomb has a periodic array of cells with hexagonal symmetry. Although the vortex medium used in this new work is a fluid rather than a solid array, it also possesses an internal set of hidden discrete symmetries. For example, one of the team's ultracold gases had the fourfold symmetry of a square, and another had the tetrahedral symmetry of a four-sided die, familiar to players of fantasy games everywhere.”
continue reading
#quantum physics#physics#condensed matter#vortex#temperature#rotation#spinning#oscillation#superconductivity#states of matter#scaling#fractal#cosmology#symmetry#energy#magnetism#tetrahedron
29 notes
·
View notes
Text

“sacred geometry: metatron’s cube, the holy 108, tetrahedron, hexahedron, octahedron, dodecahedron, isocohedron”
#screenshot#my screenshots#thoughts#ideas#sacred geometry#metatronscube#the holy 108#metatron’s cube#tetrahedron
4 notes
·
View notes