#Hemachatus
Text

Rinkhals aka Ring-necked Spitting Cobra (Hemachatus haemachatus), family Elapidae, Long Tom Pass, South Africa
Venomous.
photograph by Tyrone Ping
150 notes
·
View notes
Note
Another ask, sorry lol.
What in your opinion is or do you think would be the worst venomous snake to keep, in terms of both danger to you and fragility of the snake?
I've personally always seen mambas and spitters as the most dangerous to you, because they're just ridiculously volatile and flighty, and then sea snakes of any kind seem just about impossible to keep in a way where they're at all happy, from what I've seen.
Don't apologize, this is another great question!
You personally could not pay me to keep a rinkhals cobra (Hemachatus haemachatus). They're not true cobras, but they combine all the worst cobra traits (spitting, super potent venom) along with an extremely obstinate personality and finicky care. They're easily stressed and can crash quickly if things aren't perfect!

They also play dead when they're upset, which is honestly a bit inconvenient with a snake their size because they're heavy and awkward even when they're cooperating.
Although they're very cute actors. Look at this poor dead snake


Rinkhals are great to see, always such beautiful and charismatic snakes, but they would be terrible pets!

Sea snakes would also be terrible, though - they're so difficult to keep that most professional aquariums can't even manage it!

176 notes
·
View notes
Text
John Zorn's "COBRA" live at The Knitting Factory
Cobra was recorded at the Knitting Factory, NYC, throughout 1992. John Zorn acted as conducter for the monthly impromptu jams, which were recorded and are dated according to month.
Jeff Buckley participated in April's recordings.
Buckley contributed voice (tenor) to track 4, "Taipan," and track 5, "D. Popylepis."
youtube
@crudecube
2 years ago (edited)
Setlist for anyone trying to find a particular artist from the Knit.
0:00 - "Hemachatus Haemachatus" (Track 1)
2:12 - "Naja Naja Atra" (Track 2)
5:52 - "Many-Banded Krait" (Track 3)
16:19 - "Taipan" (Track 4)
17:49 - "D. Popylepis" (Track 5)
18:29 - "Lampropeltis Doliata Syspila" (Track 6)
21:01 - "Boomslang" (Track 7)
32:08 - "Maticora Intestinalis" (Track 8)
38:21 - "Acanthopis Antarcticus" (Track 9)
42:21 - "Hydrophiidae" (Track 10) > (Abrupt cutoff @ 44:51) ================[CUTOFF]=================
I may have got some wrong since they're so difficult to distinguish, with the ambient noise and faded transitions and all. If my approximations are correct, that leaves 4 tracks missing, which would get it to about an hour in length.
How do you play John Zorn's COBRA
The BBC documentary series On the Edge: Improvisation in Music (1992) includes a short segment of Zorn explaining and conducting two versions of Cobra.[8] His goal with Cobra, Zorn said, was to "harness" the creative developments in improvisation and extended techniques by New York City's downtown scene musicians in a semi-structured way, but "without hindering" their performances; he was interested in telling the musicians when to play, and with whom, but without telling them what to play. Plus-Minus (1963, 1974) by German composer Karlheinz Stockhausen was a key inspiration for Zorn, inspiring him to develop methods play with or against each other and in response to his cues but without dictating specific notes, sounds, or other formal structures. Though Cobra can be performed by any number of musicians plus a prompter who handles the cards, Zorn has stated that at least ten musicians are ideal, with care taken in selecting the musicians based on their improvisational skills and personalities.[7]


Release Date 1992
Duration 01:00:50
Recording Date 1992
Recording Location Knitting Factory
Jeff Buckley Guest Artist, Tenor (Vocal)
ALL MUSIC REVIEW
Live at the Knitting Factory Review by Scott Yanow
Calling this set of performances bizarre would be an understatement. John Zorn inspired (through obscure game playing that is not explained anywhere on this CD) these 14 eccentric "tributes" to different types of cobras. Because many of the performances utilize samplers and voices (in addition to conventional instruments and miscellaneous devices), the wide range of sounds attained from the 87 musicians (heard in different combinations) is impressive, if often quite unlistenable, ranging from humorous interludes to very obnoxious noise. For a few examples, "Cobra 4" has a man screaming over and over again, "Cobra 2" features a sound collage with a male opera singer repeating the same four notes continuously, and "Cobra 5" has, among its many vocal noises, a man imitating a dog barking. There are some colorful segments, but in general, these self-indulgent performances would be much more interesting to see in person than to hear on record. Taken purely as a listening experience, one is surprised that this material has even been released.


youtube
JOHN ZORN'S COBRA LIVE AT THE KNITTING FACTORY
Knitting Factory Works
release date: 1995
CD#: KFW 124
Cobra was recorded at the Knitting Factory, NYC, throughout 1992. John Zorn acted as conducter for the monthly impromptu jams, which were recorded and are dated according to month. Jeff Buckley participated in April's recordings.
Buckley contributed voice (tenor) to track 4, "Taipan," and track 5, "D. Popylepis."
Credits for both track 4 & track 5:
Jeff Buckley (voice, tenor)
M. Doughty (voice, tenor)
Judy Dunaway (voice, mezzo-soprano)
Mark Ettinger (voice, tenor)
Gisburg (voice, soprano)
Cassie Hoffman (voice, soprano)
Nina Mankin (voice, mezzo-soprano)
Chris Nelson (voice, baritone)
Juliet Palmer (voice, alto)
Wilbur Pauley (voice, bass)
Rick Porterfield (voice, baritone)
Eric Qin (voice, baritone)
#jeff buckley#john zorn#cobra#avant garde#structured improvisation#modern composition#music review#live at the knitting factory#1992#1991#1995#90s#Youtube
1 note
·
View note
Text
Even more species discovered by the reptile man
1487. Gehyra hangayi Hoser, 2018
1488. Geocrinia brettbarnetti Hoser, 2020
1489. Geocrinia brianbarnetti Hoser, 2020
1490. Geocrinia grampiansensis Hoser, 2020
1491. Geocrinia logani Hoser, 2020
1492. Geocrinia otwaysensis Hoser, 2020
1493. Geoemyda daranini Hoser, 2021
1494. Gerrhopilus carolinehoserae Hoser, 2012
1495. Gerrhopilus carolinehoserae Hoser, 2012
1496. Gerrhopilus carolinehoserae Hoser, 2013
1497. Glaphyromorphus latusumbra Hoser, 2022
1498. Glaphyromorphus nigreopunctata Hoser, 2022
1499. Gnypetoscincus smythi Hoser, 2016
1500. Goldsteintyphlops kirnerae Hoser, 2012
1501. Goldsteintyphlops wellingtoni Hoser, 2012
1502. Graptemys aurantiacooculus Hoser, 2021
1503. Graptemys brunneisoculus Hoser, 2021
1504. Graptemys flavooculus Hoser, 2021
1505. Gymnobelideus martinekae Hoser, 2018
1506. Hapsidophrys daranini Hoser, 2013
1507. Hapsidophrys pintaudii Hoser, 2013
1508. Hemachatus macconchiei Hoser, 2013
1509. Heosemys malayensis Hoser, 2021
1510. Heosemys turneri Hoser, 2021
1511. Heptathyra marcdorsei Hoser, 2021
1512. Heteronotia arcerii Hoser, 2022
1513. Heteronotia binghami Hoser, 2022
1514. Heteronotia crottyi Hoser, 2022
1515. Heteronotia grooteensis Hoser, 2022
1516. Heteronotia hoserae Hoser, 2022
1517. Heteronotia insularis Hoser, 2022
1518. Heteronotia jameswhybrowi Hoser, 2022
1519. Heteronotia keilleri Hoser, 2022
1520. Heteronotia maxinehoserae Hoser, 2022
1521. Heteronotia nonidem Hoser, 2022
1522. Heteronotia oxyi Hoser, 2022
1523. Heteronotia pailsi Hoser, 2022
1524. Heteronotia ruffellae Hoser, 2022
1525. Heteronotia sundayensis Hoser, 2022
1526. Heteronotia whybrowi Hoser, 2022
1527. Hieremys mekongensis Hoser, 2021
1528. Homopus bloemfonteinensis Hoser, 2021
1529. Homopus knysaensis Hoser, 2021
1530. Homopus trevorhawkeswoodi Hoser, 2021
1531. Honlamopus honlami Hoser, 2022
1532. Honlamopus megleesae Hoser, 2017
1533. Hoplocephalus boutrosi Hoser, 2016
1534. Hoplocephalus gedyei Hoser, 2016
1535. Hoserranae shaunwhitei Hoser, 2020
1536. Hosmeria shireensbogensis Hoser, 2020
1537. Hosmeria shuddafakup Hoser, 2020
1538. Hulimkai divergens Hoser, 2018
1539. Hulimkai ruber Hoser, 2018
1540. Hydromedusa meyeyouchelys Hoser, 2021
1541. Hydrosaurus kaup, 1828 alburyi Hoser, 2014
1542. Hypsilurus ruivenkamporum Hoser, 2016
1543. Incertanura cuspis Hoser, 2020
1544. Incertanura fakfakensis Hoser, 2020
41
41
1545. Infigo jackyhoserae Hoser, 2021
1546. Inlustanura inluster Hoser, 2020
1547. Innocuascincus dorsalux Hoser, 2022
1548. Innocuascincus piperlateralis Hoser, 2022
1549. Intellagama wellsandwellingtonorum Hoser, 2020
1550. Ipsofactoscincus davemerceicai Hoser, 2022
1551. Isopachys rosswellingtoni Hoser, 2020
1552. Jackyhosergecko jackyhoserae Hoser, 2018
1553. Jackyhoserhylea ernieswilei Hoser, 2020
1554. Jackyhoserhylea jackyhoserae Hoser, 2020
1555. Jackyhosersaur jackyhoserae Hoser, 2020
1556. Jackyhosertyphlops adelynhoserae Hoser, 2013
1557. Jackyhosertyphlops cliffrosswellingtoni Hoser, 2013
1558. Kankanophryne katrinahoserae Hoser, 2020
1559. Kankanophryne marcdorsei Hoser, 2020
1560. Kankanophryne maxinehoserae Hoser, 2020
1561. Katrinahoserserpenea bobbottomi Hoser, 2020
1562. Katrinahoserserpenea danielmannixi Hoser, 2020
1563. Katrinahoserserpenea daranini Hoser, 2020
1564. Katrinahoserserpenea evanwhittoni Hoser, 2020
1565. Katrinahoserserpenea mcconnachiei Hoser, 2020
1566. Katrinahoserserpenea rodneykingi Hoser, 2020
1567. Katrinus jackyae Hoser, 2003
1568. Kinixys divergentens Hoser, 2021
1569. Kinixys varians Hoser, 2021
1570. Kinosternon grantturneri Hoser, 2021
1571. Kullandergekko rosswellingtoni Hoser, 2021
1572. Kullandergekko wennigae Hoser, 2021
1573. Kumanjayiwalkerus kumanjayi Hoser, 2020
1574. Laemanctus tuxtlasensis Hoser, 2015
1575. Laemanctus viridis Hoser, 2015
1576. Lampropholis oxyi Hoser, 2022
1577. Lateratenebriscincus acrilineata Hoser, 2019
1578. Lateratenebriscincus albaaudere Hoser, 2019
1579. Lateratenebriscincus albavarietata Hoser, 2019
1580. Lateratenebriscincus etfatubrunnea Hoser, 2019
1581. Lateratenebriscincus freshsweetpotato Hoser, 2019
1582. Lateratenebriscincus gulagorum Hoser, 2019
1583. Lateratenebriscincus laterafusca Hoser, 2019
1584. Lateratenebriscincus leucolabialis Hoser, 2019
1585. Lateratenebriscincus maculaoccipitalis Hoser, 2019
1586. Lateratenebriscincus sentaniensis Hoser, 2019
1587. Lateratenebriscincus sepikensis Hoser, 2019
1588. Lateratenebriscincus tokpisinensis Hoser, 2019
1589. Leiopython barkeri Hoser, 2000
1590. Leiopython bennetti Hoser, 2000
1591. Leiopython hoserae Hoser, 2000
1592. Liasis cyrilhoseri Hoser, 2015
1593. Liasis sharonhoserae Hoser, 2015
1594. Libertadictus (Robinwitttyphlops) jackyhoserae Hoser, 2013
1595. Libertadictus (Slopptyphlops) richardwellsi Hoser, 2013
1596. Libertadictus (Suewitttyphlops) sloppi Hoser, 2013
1597. Limnodynastes alexantenori Hoser, 2020
1598. Limnodynastes cameronganti Hoser, 2020
1599. Limnodynastes shanescarffi Hoser, 2020
1600. Liopeltis borneoiensis Hoser, 2013
1601. Liopeltis brummeri Hoser, 2013
1602. Liopeltis philippinesiensis Hoser, 2013
42
42
1603. Liopholis dannygoodwini Hoser, 2018
1604. Litotescincus colinrayi Hoser, 2022
1605. Litotescincus evanwhittoni Hoser, 2022
1606. Litotescincus fiacummingae Hoser, 2022
1607. Litotescincus martinekae Hoser, 2022
1608. Litotescincus wellsi Hoser, 2016
1609. Llewellynura fukker Hoser, 2020
1610. Llewellynura yehbwudda Hoser, 2020
1611. Lobulia oliveetfatua Hoser, 2019
1612. Lophognathus wellingtoni Hoser, 2015
1613. Loveridgelaps fiacummingae Hoser, 2016
1614. Loveridgelaps josephburkei Hoser, 2016
1615. Loveridgelaps sloppi Hoser, 2016
1616. Loveridgelaps yeomansi Hoser, 2016
1617. Lycophidion woolfi Hoser, 2013
1618. Macmillanus jackyhoserae Hoser, 2012
1619. Macquaria honlami Hoser, 2020
1620. Macquaria hoserae Hoser, 2020
1621. Macrocalamus wellingtoni Hoser, 2020
1622. Macrocalamus wellsei Hoser, 2020
1623. Macrocerastes funki Hoser, 2013
1624. Macrocerastes hoserae Hoser, 2013
1625. Macrocerastes wellingtoni Hoser, 2013
1626. Macrocerastes wellsi Hoser, 2013
1627. Macrochelys maxhoseri Hoser, 2013
1628. Macrochelys muscati Hoser, 2013
1629. Macropogonomys aplini Hoser, 2020
1630. Macropogonomys blacki Hoser, 2020
1631. Macropogonomys gedyei Hoser, 2020
1632. Macropogonomys maxhoseri Hoser, 2020
1633. Macropogonomys mickpughi Hoser, 2020
1634. Macropogonomys mippughae Hoser, 2020
1635. Madascincus nosymangabeensis Hoser, 2015
1636. Mahonabatrachus chriswilliamsi Hoser, 2020
1637. Mahonabatrachus marionanstisae Hoser, 2020
1638. Mahonabatrachus pailsae Hoser, 2020
1639. Mahonabatrachus roypailsi Hoser, 2020
1640. Malayocolotes cassandracampbellae Hoser, 2019
1641. Mannixsaurus matthingleyi Hoser, 2018
1642. Marlenegecko (Marlenegecko) shireenhoserae Hoser, 2017
1643. Marlenegecko davidcharitoni Hoser, 2017
1644. Marlenegecko eungellaensis Hoser, 2017
1645. Matteoea dorosioi Hoser, 2020
1646. Matteoea matteoae Hoser, 2020
1647. Matteoea sommerichi Hoser, 2020
1648. Maxinehoserranae brettbarnetti Hoser, 2020
1649. Maxinehoserranae maxinehoserae Hoser, 2020
1650. Maxinehoserranae piersoni Hoser, 2020
1651. Mcphieus hadynmcphiei Hoser, 2022
1652. Mcphieus michaelmatheri Hoser, 2022
1653. Mcphieus scottgranti Hoser, 2022
1654. Megaerophis masalbidus Hoser, 2018
1655. Megaerophis promontoriumrursus Hoser, 2018
1656. Metacrinia bettyswileae Hoser, 2020
1657. Metacrinia wilhelminahughesae Hoser, 2020
1658. Mitrolysis (Invisibiliaauris) flavoranae Hoser, 2020
1659. Mitrolysis (Invisibiliaauris) leucodorsalinea Hoser, 2020
1660. Mitrolysis (Paramitrolysis) inornata Hoser, 2020
43
43
1661. Mitrolysis dumptrashensis Hoser, 2020
1662. Mixophyes (Quasimixophyes) hoserae Hoser, 2020
1663. Mixophyes couperi Hoser, 2016
1664. Mixophyes shireenae Hoser, 2016
1665. Moechaeanura (Telaater) spica Hoser, 2020
1666. Moechaeanura albatermacula Hoser, 2020
1667. Moechaeanura tritong Hoser, 2020
1668. Moloch browni Hoser, 2022
1669. Moloch granti Hoser, 2022
1670. Montivipera europa Hoser, 2016
1671. Montivipera snakebustersorum Hoser, 2016
1672. Montivipera yeomansi Hoser, 2016
1673. Morelia gray, 1842 cliffrosswellingtoni Hoser, 2018
1674. Morelia gray, 1842 harrisoni Hoser, 2000
1675. Morelia gray, 1842 macburniei Hoser, 2003
1676. Morelia gray, 1842 mippughae Hoser, 2003
1677. Morelia gray, 1842 wellsi Hoser, 2012
1678. Mosleyia (Amnisrana) michaelsmythi Hoser, 2020
1679. Mosleyia (Amnisrana) pilloti Hoser, 2020
1680. Mosleyia cottoni Hoser, 2020
1681. Motteramus floresensis Hoser, 2022
1682. Naja latebras Hoser, 2022
1683. Naja reduci Hoser, 2022
1684. Narophis cliffrosswellingtoni Hoser, 2020
1685. Narophis richardwellsei Hoser, 2020
1686. Nebulifera merceicai Hoser, 2017
1687. Neophractops rosea Hoser, 2020
1688. Nephrurus (Nephrurus) bulliardi Hoser, 2016
1689. Nephrurus (Quazinephrurus) coreyrentoni Hoser, 2016
1690. Nephrurus (Quazinephrurus) ianrentoni Hoser, 2016
1691. Nephrurus blacki Hoser, 2016
1692. Nephrurus kimberleyae Hoser, 2016
1693. Nephrurus saxacola Hoser, 2016
1694. Notanemoia cathysonnemannae Hoser, 2019
1695. Notanemoia georgemariolisi Hoser, 2019
1696. Notanemoia karlagambellae Hoser, 2019
1697. Notanemoia neilsonnemanni Hoser, 2019
1698. Nothoserphus mirabilis Brues, 1940
1699. Notopseudonaja rosswellingtoni Hoser, 2022
1700. Notopseudonaja wellsi Hoser, 2013
1701. Notoscincus fereflecka Hoser, 2022
1702. Notoscincus flecka Hoser, 2022
1703. Notoscincus monodorsa Hoser, 2022
1704. Notoscincus whoa Hoser, 2022
1705. Nyctimystes (Asperohyla) aspera Hoser, 2020
1706. Nyctimystes (Asperohyla) georgefloydi Hoser, 2020
1707. Nyctimystes (Magnummanibus) charlottae Hoser, 2020
1708. Nyctimystes (Magnummanibus) doggettae Hoser, 2020
1709. Nyctimystes mondoensis Hoser, 2020
1710. Obscuraskinkus (Sparsuskinkus) divergens Hoser, 2022
1711. Obscuraskinkus neglectus Hoser, 2022
1712. Ocellatalbum alexdudleyi Hoser, 2022
1713. Ocellatalbum assangei Hoser, 2022
1714. Ocellatalbum dannygoodwini Hoser, 2022
1715. Odatria bennymcnamarai Hoser, 2022
1716. Odatria darrenkeilleri Hoser, 2022
1717. Odatria davidhancocki Hoser, 2018
1718. Odatria funki Hoser, 2014
44
44
1719. Odatria hawkeswoodi Hoser, 2013
1720. Odatria honlami Hoser, 2013
1721. Odatria hoserae Hoser, 2013
1722. Odatria jimgreenwoodi Hoser, 2018
1723. Odatria maderi Hoser, 2014
1724. Odatria nini Hoser, 2013
1725. Odatria sawuensis Hoser, 2022
1726. Oedura bulliardi Hoser, 2017
1727. Oedura rentonorum Hoser, 2017
1728. Oedura whartoni Hoser, 2017
1729. Oedurella (Parvusdactylus) alba Hoser, 2017
1730. Oedurella (Parvusdactylus) garystephensoni Hoser, 2017
1731. Oedurella (Parvusdactylus) jamielindi Hoser, 2017
1732. Oedurella (Parvusdactylus) sonnemanni Hoser, 2017
1733. Oedurella minima Hoser, 2017
1734. Oopholis adelynhoserae Hoser, 2012
1735. Oopholis jackyhoserae Hoser, 2012
1736. Oopholis oxyi Hoser, 2018
1737. Opacitascincus ugh Hoser, 2022
1738. Ophiomorus macconchiei Hoser, 2015
1739. Ophioscincus paulwoolfi Hoser, 2020
1740. Oreocalamus turneri Hoser, 2020
1741. Orlitia perakensis Hoser, 2021
1742. Ornatanura leucopicturas Hoser, 2020
1743. Ornatanura parscinereo Hoser, 2020
1744. Ornatanura parsviridis Hoser, 2020
1745. Oxychelys oxyi Hoser, 2021
1746. Oxyuranus adelynhoserae Hoser, 2009
1747. Oxyuranus andrewwilsoni Hoser, 2009
1748. Oxyuranus barringeri Hoser, 2002
1749. Pailsus hoserae Hoser, 2013
1750. Pailsus pailsei Hoser, 1998
1751. Pailsus rossignollii Hoser, 2000
1752. Panacedechis trevorhawkeswoodi Hoser, 2009
1753. Pantherosaurus bulliardi Hoser, 2015
1754. Pantherosaurus maxhoseri Hoser, 2015
1755. Pantherosaurus queenslandensis Hoser, 2015
1756. Paracrinia funki Hoser, 2020
1757. Paracrinia lenhoseri Hoser, 2020
1758. Paramixophyes piersoni Hoser, 2016
1759. Paramixophyes yeomansi Hoser, 2016
1760. Pareas malayensis Hoser, 2016
1761. Pareas sumatrensis Hoser, 2016
1762. Parvavaranus apicemalba Hoser, 2020
1763. Parvavaranus ignis Hoser, 2020
1764. Parvavaranus pyrrhus Hoser, 2020
1765. Pelleyus pelleyi Hoser, 2015
1766. Pelleyus punjabensis Hoser, 2015
1767. Pelochelys clivepalmeri Hoser, 2014
1768. Pelochelys telstraorum Hoser, 2014
1769. Pelomedusa alexstaszewskii Hoser, 2021
1770. Pelomedusa dannygoodwini Hoser, 2021
1771. Pelomedusa darrenkeilleri Hoser, 2021
1772. Pelomedusa shannonmcgrathi Hoser, 2021
1773. Pelusios divergentans Hoser, 2021
1774. Pelusios lynnrawi Hoser, 2021
1775. Petaurus adelynhoserae Hoser, 2020
1776. Petrogale hoserae Hoser, 2020
45
45
1777. Petrogale martinekae Hoser, 2020
1778. Petrogale ordensis Hoser, 2020
1779. Petrogale pentecostensis Hoser, 2020
1780. Philippinedraco brunneis Hoser, 2022
1781. Philippinedraco graysoni Hoser, 2022
1782. Philippinedraco toscanoi Hoser, 2022
1783. Philocryphus hoserae Hoser, 2019
1784. Phryia paulhorneri Hoser, 2018
1785. Piersonus bartletti Hoser, 2020
1786. Pilgerus assangei Hoser, 2014
1787. Pilgerus macki Hoser, 2014
1788. Pilgerus mooreae Hoser, 2014
1789. Pilgerus nardellai Hoser, 2014
1790. Platyplectron gerrymarantellii Hoser, 2020
1791. Platyplectron snakemansbogensis Hoser, 2020
1792. Platyplectron timjamesi Hoser, 2020
1793. Platyplectrum shaneblacki Hoser, 2016
1794. Pogonomys sharonhoserae Hoser, 2020
1795. Pointednasus (Viridihaema) extentadigitus Hoser, 2019
1796. Pointednasus (Viridihaema) flavopalpebrae Hoser, 2019
1797. Pointednasus (Viridihaema) labiamarmorata Hoser, 2019
1798. Pointednasus (Viridihaema) makiraensis Hoser, 2019
1799. Pointednasus (Viridihaema) ventriiridescens Hoser, 2019
1800. Pointednasus clavoflavoviridis Hoser, 2019
1801. Pointednasus currearbor Hoser, 2019
1802. Pointednasus flavorecta Hoser, 2019
1803. Pointednasus widerecta Hoser, 2019
1804. Potorous waddahyamin Hoser, 2020
1805. Proablepharus absconditus Hoser, 2022
1806. Proablepharus alexanderdudleyi Hoser, 2022
1807. Proablepharus elonginquo Hoser, 2022
1808. Proablepharus eughr Hoser, 2022
1809. Proablepharus garystephensoni Hoser, 2022
1810. Proablepharus igh Hoser, 2022
1811. Proablepharus insolitum Hoser, 2022
1812. Proablepharus jessicabriggsae Hoser, 2022
1813. Proablepharus martinekae Hoser, 2022
1814. Proablepharus micra Hoser, 2022
1815. Proablepharus stevebennetti Hoser, 2022
1816. Pseudechis eipperi Hoser, 2003
1817. Pseudechis rentoni Hoser, 2003
1818. Pseudemoia danielmannixi Hoser, 2022
1819. Pseudemoia davidkerryi Hoser, 2022
1820. Pseudemoia mcnamarai Hoser, 2022
1821. Pseudochirops (Sloppossum) chrismaxwelli Hoser, 2020
1822. Pseudochirops fiacummingae Hoser, 2020
1823. Pseudochirops jamesbondi Hoser, 2020
1824. Pseudochirops waddamaddawidyu Hoser, 2020
1825. Pseudodelma cummingae Hoser, 2017
1826. Pseudomys (Farkmys) albapes Hoser, 2020
1827. Pseudomys (Ohmys) griseorursus Hoser, 2020
1828. Pseudomys (Ohmys) pesrosea Hoser, 2020
1829. Pseudomys (Oimys) pellicauda Hoser, 2020
1830. Pseudomys (Ouchmys) luxauris Hoser, 2020
1831. Pseudomys (Ouchmys) occultatum Hoser, 2020
1832. Pseudonaja charlespiersoni Hoser, 2009
1833. Pseudonaja cliveevattii Hoser, 2009
1834. Pseudonaja elliotti Hoser, 2003
0 notes
Photo

(Hemachatus haemachatus) rinkhals
This snake has a few tricks up its sleeve. If shooting streams of venom at the eyes of potential threats is an ineffective deterrent, the rinkhals will play dead before finally biting as a last string defense.
#snake#nature#earth#reptile#venomous#animals#Hemachatus haemachatus#Hemachatus#rinkhals#spitting cobra
2K notes
·
View notes
Text

Rinkhals - Hemachatus haemachatus
Grahamstown, Eastern Cape, South Africa
4 notes
·
View notes
Text
Herp of the day: Rinkhals

Rinkhals, or the Ring Necked Spitting Cobra (Hemachatus haemachatus) is a venomous snake species from Africa.
Although it looks similar to a cobra, they are not, these spitting snakes can spray its venom up to 2.5 m. Its spitting mechanism is primitive and it has to rear up and fling its body forward to spray its venom.
It is also known to fake death by rolling onto its back with its mouth agape. The rinkhals has a varied diet, its main prey is toads, but it also eats small mammals, amphibians, and other reptiles.
#herpoftheday#snake#snakes#Rinkhals#Rinkhal#venomous#elapid#elapidae#herps#herp#herpetology#biology#animals#wildlife#reptile#reptiles#Hemachatus#Hemachatushaemachatus#Hemachatus haemachatus
24 notes
·
View notes
Text
Did snakes evolve to spit venom in the eyes of humans?
Some snakes can spit venom.
Now, this is the sort of thing that I assume is common knowledge, because I assume that everyone went through a phase at age 8 where they read obsessively about snakes and other reptiles after discovering that libraries exist.
What’s that? This isn’t a universal experience? Wild :0
Well, if you weren’t a deeply weird 8-year-old child who likes snakes a lot, then you get TWO fun facts this week!
First, there are snakes that can spit venom. This is kind of crazy, because generally, venom is all about close-contact. if the toxin is delivered via a bite, it’s venom. otherwise, it’s poison. Right? Right?!
well... things can get complicated when the venom is being shot out of the tooth.

(A slightly more correct way to think of venomous-vs-poisonous is whether the toxin delivery is active or passive. If the animal as actively trying to put the toxin in you, it’s venomous. If you get the toxin in you without the animal needing to do anything, then it’s poisonous. Probably there are exceptions to this, because nature LOVES an exception, but that’s the basic gist of it)
There’s only a handful of species that can spit venom, all of which are cobras. That said, it appears that this trait evolved at least 3 different times: in Asian spitting cobras (Naja, subgenus Naja), African spitting cobras (Naja, subgenus Afronaja), and Rinkhals (Hemachatus; yes I’m counting rinkhals as cobras, because i think they deserve it).
How do snakes spit venom? Basically, they flex their head and neck really hard and shoot the venom out of a little hole in the front of their fangs, which are modified from the standard cobra fang venom-delivery-system:

the small round hole (& the angle leading to the hole) lets them spew their venom up to 2.5 meters. And to be clear: they DO aim for the eyes. Which: rude. But also: fair.
To make it even more interesting, the venom of spitting cobras is an altered version of the standard cobra venom recipe (which was already deadly) which is EVEN MORE PAINFUL. Ow.
Okay, so that’s the basics about snakes that spit venom.
Fun fact number 2: these snakes (maybe) evolved this venom spitting ability specifically to spit in the eyes of humans. Or, hominins, at least.
Why do we think this? Well, first of all, there’s the fact that it seems like this ability evolved 3 different times (at least). Rather than one species developing a venom-spitting ability, and then evolved into the different species we see today, it appears that there were 3 different times where the ability arose:
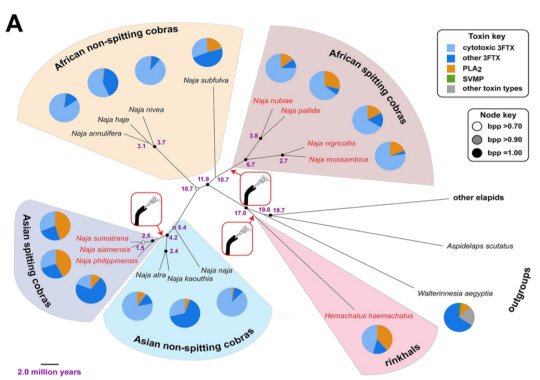
this is what we call convergent evolution. (This diagram is kind of a lot, so I over-simplified it into tree on ms paint. This diagram is NOT to-scale with the time line)
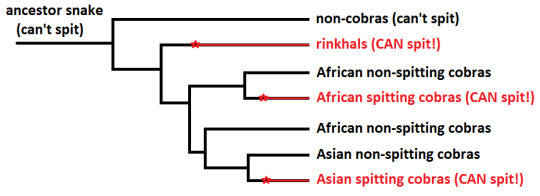
The stars are where we think the ability to spit venom arose (again: not to scale). There is actually a good amount of evidence to support the 3 different origins of this ability (which can be read about here or here).
Generally, convergent evolution happens when species are exposed to the same sorts of environments/ predators/ threats. Sometimes, a problem has one solution that just works. Dolphins and sharks look similar because they found a really good design for cruising in the ocean.
So, what problem does spitting venom solve?
Well. Around the time these adaptations appeared, there were these weird apes that started walking around on two legs and, importantly, using tools that let them attack snakes from a distance. They could extend their reach with altered sticks, or throw rocks, or tie a rock to a stick and throw that, and when the snakes bit these weapons, nothing happened, because the predator was over there, just out of reach.

Problem: the weird ape predator is too far away to bite & envenomate
Solution: throw the venom in their eyes.
(to be clear, the weird apes are hominins)
Now, this isn’t 100% certain, because palaeontology and anthropology are notoriously difficult fields in which to prove things, on account of the lack of a time machine. (seriously, the academic fights in palaeontology can get spicy).
BUT scientists noted that a) spitting venom is particularly effective against humans, and b) the ability to spit venom seems to have appeared not long after bipedal hominins arrived in their areas.
African spitting cobras: evolved ~6.7 million years ago.
Hominins (genus Pan): appeared in the area ~7 million years ago.
Asian spitting cobras: evolved ~2.5 million years ago.
Hominins (Homo erectus): appeared in the area ~2.5 million years ago.

(since rinkhals branched off so long ago that we don’t really have a good idea of how long ago they evolved the ability to spit venom; they split off from other cobras ~17 MYA, but their ability to spit venom could have arisen at any point since then. Monotypic genera are like that sometimes (ಥ_ಥ). Genetic studies pending while we Figure Shit Out)
Is it possible that the spitting venom evolved to deal with something else? Yes, it is possible. But, with the evidence we have currently, this is a hypothesis with a reasonable amount of support.
Also, I just think it’s delightful to think of snakes evolving to spit in my eye.

#biology#fun facts#fun fact friday#Herpetology#zoology#snakes#STEM#adhd in STEM#science#sciblr#science side of tumblr#ecology#cobra
530 notes
·
View notes
Text
Netcare 911 paramedic and professional snake handler rescues multiple snakes from properties around the Vaal Triangle
Netcare 911 paramedic and professional snake handler rescues multiple snakes from properties around the Vaal Triangle
Gauteng: Netcare 911 paramedic Nick van Der Walt, also a professional snake handler was asked to remove snakes from properties in Vanderbijlpark and Midvaal. After multiple days of captures in the extreme heat, all Nick’s rescues were Rinkhals snakes (Hemachatus haemachatus) One of the many traits of the Rinkhals is to play dead with its…Netcare 911 paramedic and professional snake handler…

View On WordPress
0 notes
Text
Spitting cobra venom didn't evolve to kill – just to put us in a world of pain
https://sciencespies.com/nature/spitting-cobra-venom-didnt-evolve-to-kill-just-to-put-us-in-a-world-of-pain/
Spitting cobra venom didn't evolve to kill – just to put us in a world of pain
Like pappy used to say: if you see it writhe and hiss, give it a miss. Humans have passed on similar nuggets of wisdom since time immemorial, giving us a healthy (and sometimes less than healthy) respect for backing away from snakes.
Yet the fact snake bites hurt is usually an accident of nature – virtually every single species of snake evolved the ability to inject venom to catch prey, rather than for defence. But there are exceptions to the rule – new research shows some snakes actually evolved their venom specifically to drive away potential predators, including us. And it’s excruciatingly painful for good reason.
The study by an international team of scientists demonstrates a unique kind of venom and envenomation strategy evolved on at least three different occasions.
Spitting cobras and rinkhals are closely related snakes capable of squirting venom far enough for an unwary human to cop an eyeful.
The effects aren’t pleasant. Chemicals in the spitting cobra’s venom chew away at the cornea, causing intense pain and – in high enough doses – blindness.
Fangs like water-pistols and blinding venom aren’t exactly suited to bringing down a quick-footed meal, so researchers saw this as a perfect opportunity to investigate the evolutionary history of this particularly unusual characteristic.
Snakes first evolved an ability to inject venom somewhere between 60 and 80 million years ago. Since then, thousands of species in the superfamily Colubroidea have tweaked the original recipe and modified their mouth bits to suit their dietary needs.
Using fossils as calibration, the researchers used molecular dating techniques on the genomes of a number of true spitting cobra (Naja species), rinkhals (Hemachatus haemachatus), and non-spitting relatives (Walterinnesia aegyptia and Aspidelaps scutatus).
The results suggested African spitting cobras developed their eye-watering habit between 6.7 and 10.7 million years ago. Their Asian relatives followed suit 4 million years later.
Rinkhals were hard to pin down but must have developed their ability since parting from other spitting cobras more than 17 million years ago.
An analysis of their venoms revealed their make-up had more in common with each other than non-spitting relatives, with the exception of the more neurotoxic recipe belonging to Naja philippinensis.
The agonising effects of the venom’s chemistry were also put to the test using living tissue and nerve samples.
“We tested how venom components affected pain-sensing nerves and showed that spitting cobra venoms are more effective at causing pain than their non-spitting counterparts,” says neuropharmacologist Irina Vetter from the University of Queensland in Australia.
Added together, the sum of the results implies spitting cobras and rinkhals repurposed their venom and fangs independently, transforming into defence mechanisms capable of detracting large predators.
And just who were those large predators in each case? We can only guess, but the researchers argue there are some good reasons to think our ancestors fit the bill, citing evidence of snakes influencing primate neurobiology and behaviour.
Humans and chimpanzees diverged roughly around the same time African spitting cobras evolved the trait. We’re also likely to identify potentially venomous snakes and attack from a short distance, putting pressure on spitting as a suitable means of venom delivery.
Of course, it’s all speculation based on loosely circumstantial evidence at this point, but the ideas warrant further exploration.
“It is intriguing to think that our ancestors may have influenced the origin of this defensive chemical weapon in snakes,” says snake venom expert Nick Casewell from the University of Liverpool.
Ironically, by understanding the evolutionary relationship between toxins and our own bodies, we’re better positioned to identify potential mechanisms for new classes of medicine.
The analgesic properties of rattlesnake venom, for example, could give millions of people living with neuropathic pain a chance of relief.
“Pain-causing toxins from animal venoms can be useful tools to help us understand pain signalling at a molecular level and are helping us to identify new targets for future painkillers,” says University of Queensland molecular biologist Sam Robinson.
It’s time we called a truce with the spitting cobra. We’ll keep our distance, and appreciate their skills from afar.
This research was published in Science.
#Nature
1 note
·
View note
Text

Ring-necked Spitting Cobra aka Rinkhals (Hemachatus haemachatus), family Elapidae, Grahamstown, South Africa
Venomous.
photograph by Tyrone James Ping
147 notes
·
View notes
Note
So...question? Why are king cobras called cobras and why do they look like them if they are not actually cobras?
Great question! The answer is basically that they just look a lot like cobras.
The only true cobras are those in the genus Naja. They all have hoods and a characteristic threat display of raising straight up, but they're also distinguished by their ability to spit venom (all true cobras except for Indian and Caspian cobras can spit) and their diet, which is opportunistic to the extreme (most will eat anything, including carrion, which is usually a no-go for most snakes). They also tend to be large-medium sized snakes, with most reaching around 6 feet long.

King cobras, Ophiophagus hannah, are very closely related to true cobras (they're kind of like a middle ground been Naja, the true cobras, and Dendroaspis, the mambas). They look a lot like cobras, including their straight-up threat display and hoods. They're distinguished from true cobras not only by their inability to spit venom but by their size (they're much longer, averaging 12 feet but sometimes reaching up to 18) and their diet, which is almost exclusively other snakes.

There are a few other snakes that are called "cobras" despite not being true cobras at all, including shield-nosed cobras and tree cobras, but the other really big one that looks like a cobra but isn't is the rinkhals cobra, Hemachatus haemachatus. They have keeled dorsal scales, unlike true cobras, and can play dead when threatened.

220 notes
·
View notes
Photo

Cobra (The Stallone's movie)
Take it only with medical prescription.
The Wikipedia says that:
Cobra is the common name of various elapid snakes, most of which belonging to the genus Naja.
All of the known cobras are venomous and many are capable of rearing upwards and producing a hood when threatened.
All members of the genus Naja, the "true" cobras, rear and produce hoods.
Other "cobra" genera and species are as follows:
The rinkhals, or ring-necked spitting cobra, Hemachatus haemachatus, so called for its neck band as well as its habit of rearing and spreading its hood when disturbed.
The king cobra, Ophiophagus hannah.
The two species of tree cobra, Goldie's tree cobra and black tree cobra
The two species of shieldnose cobras, cape coral snake and Aspidelaps scutatus.
The two species of black desert cobra, desert black snake and Walterinnesia morgani, neither of which rears or produces a hood.
The so-called American cobra, the Eastern coral snake, which also does not rear or produce a hood.
The false water cobra, Hydrodynastes gigas, is the only "cobra" that is not a member of the Elapidae. It does not rear, produces only a slight flattening of the neck, and is only mildly venomous.
#Javier L#superColoroid#superColoroidDrawings#drawings#drawing#digital#art#ink#MangaStudio#color#movie#disease#cure#The strong arm of the law#aspirin#Stallone#sylvester stallone
1 note
·
View note
Photo

(Hemachatus haemachatus) rinkhals
This snake’s venom is a neuro/cytotoxic cocktail that is typically shot directly into the eyes. This Elapid is a “false cobra”, as it does not belong to the genus, Naja, and is known to play dead.
#snake#nature#earth#reptile#animals#venomous#Elapidae#Elapid#Hemachatus haemachatus#rinkhals#Hemachatus#ring-necked spitting cobra
109 notes
·
View notes
Photo

The Rinkhals(Hemachatus haemachatus) is native to the South-Africa and is often mistaken for other snake species. via /r/Awwducational http://ift.tt/2pHgUYa: http://ift.tt/2pHBRTb
0 notes
Photo

Faala galera blz? Galera essa ai e a HEMACHATUS HAEMACHATUS ou como é popularmente conhecida NAJA CUSPIDEIRA MOÇAMBICANA Reino:Animalia Filo:Chordata Subfilo:Vertebrata Classe:Reptilia Ordem:Squamata Subordem:Serpentes Família:Elapidae Género:Haemachatus Espécie:H. Haemachatus Hemachatus haemachatus ou naja cuspideira moçambicana é uma espécie de serpente que pode medir de 1 a 3 metros, habita savanas úmidas, pastagens e florestas. Alimenta-se de roedores pequenos, aves, lagartos e outras cobras menores. Possui neurotoxina que paralisa o sistema nervoso causando parada respiratória, levando à morte. Pode ser encontrada na África do Sul. Pode ter o corpo preto ou marrom escuro com um anel branco no pescoço. Tem vários mecanismos de defesa: pode dilatar seu pescoço na expansão que é chamada de "capuz", que faz com que pareça maior e mais ameaçadora; pode morder e injetar veneno na vítima atacada; pode lançar veneno nos olhos de seu inimigo. São predadores fantásticos e engenhosos mas são vulneráveis a ataques. A forma de lançar o veneno é a seguinte: os dutos que conduzem os venenos são fechados e existe um buraco na parte da frente diante de cada dente, quando a naja comprime esses sacos de veneno os dentes atuam como pistolas de água, esguichando veneno a uma distância de quase 4 metros, em geral nos olhos do inimigo, uma arma que pode ser usada tanto para defesa como no ataque. #TodoDiaAlgoNovo https://www.instagram.com/p/Bqpwg0YHYqL/?utm_source=ig_tumblr_share&igshid=itzokxq94158
0 notes