#stoichiometry
Text
in evil chemistry, the equation balances you
#can you tell i’m tired#i’m so tired#fuck balancing equations man#why is it so awful for me#like i can d o it (sometimes)#it’s just horrifically strenuous and painful#chemistry#science#chemistry meme#chemistry student#balancing equations#stoichiometry#god.#science side of tumblr
89 notes
·
View notes
Text
it’s almost 3 am and i’m screaming about moles & stoichiometry but in a giddy fun way because like FINALLY, something i actually understand and enjoy learning from the subject
#i’ve missed her organic chem hates my guts i swear#but this however#it’s so familiar to me asjkld#maybe i’m being weird but#and maybe the sleep deprivation has gotten to me#but you know!!#(actually i’m kinda scared i just jinxed it because i’ve actually only finished one example question 😭 and i’m already this happy lmao#you’d think i had just finished the entire page)#but okay anyways#stoichiometry#chemistry#chemblr#that name is funny to me but in a good affectionate way#nadirants
17 notes
·
View notes
Text
im going to cry
4 notes
·
View notes
Text
The Chemistry That Surround Us: Stoichiometry

2 notes
·
View notes
Text
WHAT THE ABSOLUTE FUCK IS THISSSSSS😭😭😭 (stoichiometry)
3 notes
·
View notes
Text
attempting to explain stoichiometry
💗hello everyone💗 stoichiometry is a concept that is crucial to chemistry, and really is used with all other sciences, but it can be difficult to understand so I hope I can try to explain <3
even if you don't end up getting the math, all you need to know is the general formula for all stoichiometry is (if your conversion values are equivalent...)
original value with given units * (conversion value but with the units you want / conversion value with given units) = original value but with the units you want
why does this work? if your conversion values are equivalent, then when you divide them by each other, it's gonna equal 1, and when you multiply something by 1, you get an equal value! moreover, when we write it like this, our original
while we apply this concept to chemical equations, you have probably been doing it intuitively your whole life, for example...
1 hour * (60 minutes / 1 hour) = (1 hour * 60 minutes)/(1 hour) = 60 minutes
at first glance, if you thought of it math-wise, you might not think that 60 minutes equals 1 hour, however we all know that there are 60 minutes in 1 hour :) we can use this kind of model to calculate more complicated
3 hours * (60 minutes / 1 hour) = (3 hours * 60 minutes)/(1 hour) = 3 * 60 minutes = 180 minutes
notice how we cancel out the units, but not the 3, because there's nothing to divide the 3 by on the bottom!
however, our conversion factors won't always be neat like this. think about at a grocery store, you can buy a combo box of 3 oranges and 2 apples for five dollars. you need 9 oranges for a recipe, and you want to know how many apples are gonna come with that because you don't need them and want to give them to a friend. since these come as a package (as in, for every 3 oranges you will get 2 apples (think back to how for every 1 hour we have 60 minutes) they will be equivalent stoichiometrically) we can write out an equation like this:
9 oranges * (2 apples / 3 oranges) = (9 oranges * 2 apples)/(3 oranges) = (9 * 2 apples)/(3) = (18 apples)/3 = 6 apples
while we probably would never do this kind of equation to solve this in real life, we are applying the same concepts when we do this kind of easy math in our head. that exact point shows how easy it is to use stoichiometry in application to chemistry, for example...
Mg + 2F -> MgF2
if we want to find how many MgF2 (magnesium fluoride) molecules we can make with say, 2 moles of magnesium (assuming we have an infinite store of fluorine), we can write this equation as...
2 moles of magnesium * (2 moles of fluorine / 1 mole of magnesium) = 4 moles of fluorine
notice how we use the same principle as the apples and oranges! for every one magnesium molecule (or in this case, 1 mole, but it doesn't matter because we're just worried about the ratio) we would need two fluorine molecules just to make the one molecule of magnesium fluoride!
I hope this helps! I can also help with your homework in the replies :D
#chemistry#stoichiometry#science#math#mathblr#help#studyblr#studying#student#stem#stemblr#study help
3 notes
·
View notes
Text

This is just a scratch of jax and pomni being forced to learn stoichiometry keep in mind this is just a sketch and stuff that I might turn digital i drew the fairy first and then thought “wouldn’t it be funny if it was this situation?” So I did this…
#tadc jax#jax#the amazing digital circus jax#jax fanart#tadc pomni#pomni#the amazing digital circus pomni#pomni fanart#i love pomni#stoichiometry#chemistry#pls help#spain without the s#i thought it was funny#the amazing digital circus#tadc fanart#tadc#the amazing digital circus fanart
5 notes
·
View notes
Text

thursday 26/05/22
je suis fighting for my LIFE
end of year exams are coming up w chem paper 1 being the first. i never understood stoichiometry and now i’m fighting for my life to do so. waking up at 3am and trying to function is surprisingly effective short term. i’ve been getting 3h of work in every day, i’ll definitely finish stoichiometry tomorrow
♫ scrambled - havelock ♫
#study aesthetic#studyblr#studyspo#chemistry#stem#women in stem#stoichiometry#why is it spelled like that#thefuck#chemspo#ib chemistry hl#ibdp#international baccalaureate#studying#aesthetic#mole concept#avogadro#ya boi avo#6.02x10^33
35 notes
·
View notes
Text
sustainable planting

Say for some fucked up reason i want 5 potatoes every day
the growth rate of potatoes is around 9 potatoes every 120 days
B=1 because alpha=beta
so (5)(1)(120)/(1)(9)
so round 67 plants needed to sustain that
Use stoichiometry when trying to self sustain numbnuts!!
0 notes
Text
A weak acid reacts incompletely with water to form less than stoichiometric amounts of H3O+.
"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
#book quote#chemistry#nonfiction#textbook#acid#weak acid#chemical reactions#water#hydronium#stoichiometry
0 notes
Text
We can see in figure 7.43 that there are also four cations within the unit cell, resulting in the correct 1 : 1 stoichiometry. In this structure, as seen in figure 7.43b, every cation is surrounded by four anions, and every anion is surrounded by four cations.
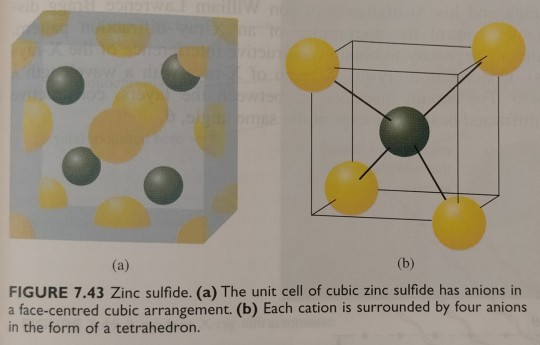
"Chemistry" 2e - Blackman, A., Bottle, S., Schmid, S., Mocerino, M., Wille, U.
#book quotes#chemistry#nonfiction#textbook#zinc sulfide#zinc#sulfur#ions#cation#anion#cubic#geometry#tetrahedron#stoichiometry
0 notes
Text
Basic concepts of Chemistry class 11
Chemistry is the scientific study of matter, its properties, and the interactions between different substances. The subject plays a crucial role in our understanding of the natural world and is essential for various fields such as medicine, engineering, and materials science. In Class 11, students are introduced to the basic concepts of chemistry, which lay the foundation for further study in the…

View On WordPress
#acidsandbases#atomicstructure#atoms#balancingchemicalequations#chemicalbonding#chemicalreactions#Class11#education#electrons#gases#liquids#molecules#protons#quantitativerelationships#science#solids#statesofmatter#stoichiometry#chemistry
1 note
·
View note
Text
Our Industrial Stoichiometry

#chemistry#science#chemistry class#education#industrial#stoichiometry#industry#manufacturing#pharmacy#agroindustry#english
0 notes
Text
Big round ass anal milf xxx Cory Chase in Revenge On Your Father
Petite brunette babe shows domination in a blind sex
Comedor sergipano e loira casada safada
Bi stud doggystyled during pussylicking
skinny twink anal fucking
Rabudo dando o cu de calcinha
Comendo novinha no escuro
Teen getting fucked up her asshole in the bathtub
Arab teen gay sex movie Hugely Hung Boys Luke And Steven
BBW phat ass takes BBC
#Limacinidae#Petofi#polyterpene#unpridefully#regenerate#planury#Meridian#koff#pachymeningitic#Josephine's-lily#half-learning#world-wept#pretypographical#stoichiometry#enamel#anality#counterphobic#Sakell#communalisation#habille
0 notes
Text
every time i engage even a little bit in Cats the musical I fall back into the hole for a little bit
#cats the musical#cats 1998#i love this fucking musical so damn much dude you have no idea#if you haven't watched it. its all on youtube. i highly highly recommend it.#its also all in a playlist in chronological order#that playlist used to be my study music dude id be jamming out to magical mister mistoffelees while doing stoichiometry#the movie was a fucking disservice and a disgrace all my homies hate the cats 2019 movie#also. gay people. in the musical. gay people real.#i could go on and on about how much i love this musical but i wont#unless...#i do understand that it's not for everyone though. it is a bit of an acquired taste (probably)#ludwig's laments
76 notes
·
View notes